Below, ESR 10, Abhinav Thareja of UoB, describes how difficult it is to get treatment to the back of the eye, due to the natural barriers. His work, recently presented at ARVO 2022, showcases how penetration enhancing agents (PEAs) can transform current treatment options for posterior segment disease.
Sneaking drugs past the eye
The very elusive road to non-invasive drug delivery via eye-drops to the posterior ocular segment for the treatment of retinal damage and disease passes through multiple twists and turns, with all the anatomical and physiological barriers of the eye acting as roadblocks resulting in very low bioavailability beyond cornea, typically less than 5%. To address this challenge, we employ penetration enhancing agents (PEAs) which present a very promising approach in topical drug delivery towards increasing the drug bioavailability in the fundus oculi by facilitating increased penetration across bio-barriers along with cellular internalization in the tissues and longer retention in the posterior segment. PEAs can be chemical or biological agents that induce an enhanced permeability of the defensive ocular barriers and membranes of cells and tissues to pharmaceutical molecules by altering their properties through a combination of known and unknown mechanisms.
Our PEAs include a novel in-house synthesized helical organic polymer and commercially available amphipathic cationic cell penetrating peptides (CPPs) that exhibit a distinct property to internalize across cellular membranes via an energy dependent or independent pathway or a combination of both, and without any need for chiral interactions with surface receptors. A lot of research has been reported on the use of CPPs in drug delivery, e.g., topical transdermal delivery, nasal delivery to pulmonary system, and delivery to the central nervous system across the blood-brain barrier, however their use in ocular drug delivery is relatively new. Read our review on PEAs for retinal drug delivery here.

So far, we have achieved an almost 15-fold increase in the transcorneal penetration of dexamethasone sodium phosphate after topical application with PEAs without causing any damage to the corneal barrier integrity or in-vitro toxicity. This can potentially increase the bioavailability of DSP in the posterior segment tissues. Our most recent work presented at ARVO 2022.
Our exquisite ex-vivo and in-vitro models
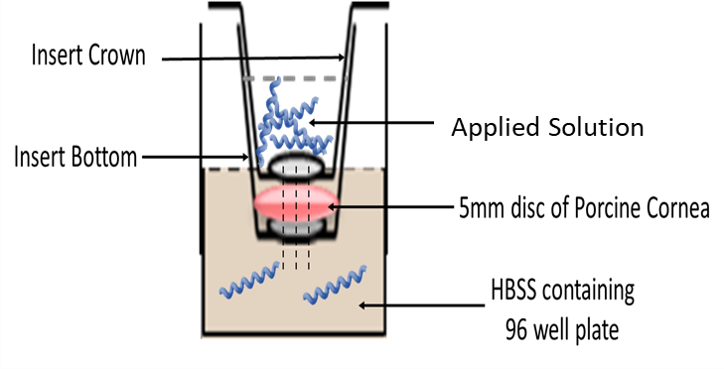
Our research group in Neuroscience and Ophthalmology at the University of Birmingham had recently developed a very reliable ex-vivo model of penetration using pig eyes which can be used to rapidly assay penetration of formulations across ocular tissues like cornea, conjunctiva, and sclera using 5mm biopsy discs from the tissue in a 96-well microplate thereby allowing multiplex measurements at the same time with different formulations rather than using the whole tissue for each measurement. This way we can maximize the tissue utility and reduce wastage while being cost-effective at the same time. This model has been validated for drug delivery.
Another very interesting model developed in our group which we extensively use in our work is an in-vitro primary neuronal cell model from rat retinas, mainly RGC neurons. It serves as an ideal model for testing tolerance of our formulations on primary neurons while the neurite outgrowths from these neurons also give a clear indication of therapeutic effect of neuroprotective formulations as a majority of neuroprotective strategies are broadly targeted towards the survival pathways of RGCs and the mechanisms of their degeneration and death in disease and can be used in multiple retinal diseases. More on this topic can be found, here.


Networking Networking
Being fond of traveling and exploring different places, travel comes complimentary with academia in the form of international conferences. Most importantly these are hotspots for networking and brilliant platforms to disseminate our research and share ideas with the best minds in the field. And so, we recently travelled to Denver in the USA to attend ARVO 2022 general congress to present our work, which happens to be the biggest ophthalmology conference globally. Besides, ORBITAL itself being a global consortium, we have been travelling across countries for workshops and trainings. Be it our first ever in-person all consortium meeting in Santiago or the last workshop on ex-vivo animal models in beautiful Parma, they have been excellent avenues for learning. We also had a very insightful world café PPI event at our Patient as a Teacher workshop in Lisbon which really helped us gain an understanding of patients’ perspective on research in complex ocular diseases and their expectations. Even more exciting is the fact that I will soon be travelling to Lithuania to Experimentica for my industrial secondment, which is an integral part of Marie Curie ITNs, where I will test my formulations in in-vivo models of ocular disease. I am really looking forward to it and more details will follow soon.


